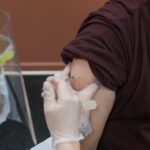
A clinical drugs trial in France has ended in tragedy after five people were hospitalised with serious neurological complications and one was left brain dead.
It has been reported that 90 people were administered the drug, which has not yet been named.
The French Health Ministry has since stopped the drugs trial and recalled all of those volunteers who took part in it.
Taken orally, the drug was undergoing a Phase I clinical trial at a licensed private European laboratory that specialises in clinical trials.
The Paris prosecutor’s office said an investigation had been opened.
French health minister, Marisol Touraine, pledged to get to the bottom of what happened.
In a message posted on its website, the company, Bio Trial insisted that “international regulations” and Bio Trial’s own procedures “were followed at every stage”.
Clinical trials: How do they work?
Drugs are often put through 3 stages before being released to the public.
Phase I: This is the first stage of the trial. A small number of people, some healthy, some with medical conditions are given a tiny dose of the drug. This is just to check whether there are any side-effects.
Phase II: The drugs effectiveness is then tested on those with medical conditions to determine whether it does actually help them.
Phase III: Drugs have to pass the first two phases before reaching this stage. They are then tested against a placebo or existing drug to see if they work.
The tragedy with this drug occurred in the first stage of the clinical trial.
Akashic Times is the UK’s only online, fully independent not-for-profit newspaper that brings you real news from across the globe.
If you want to keep ahead of what is really going on in the world, subscribe to our newspaper via the subscribe button and join our Facebook & Twitter pages. Subscription is completely free ofcourse














Follow Us!